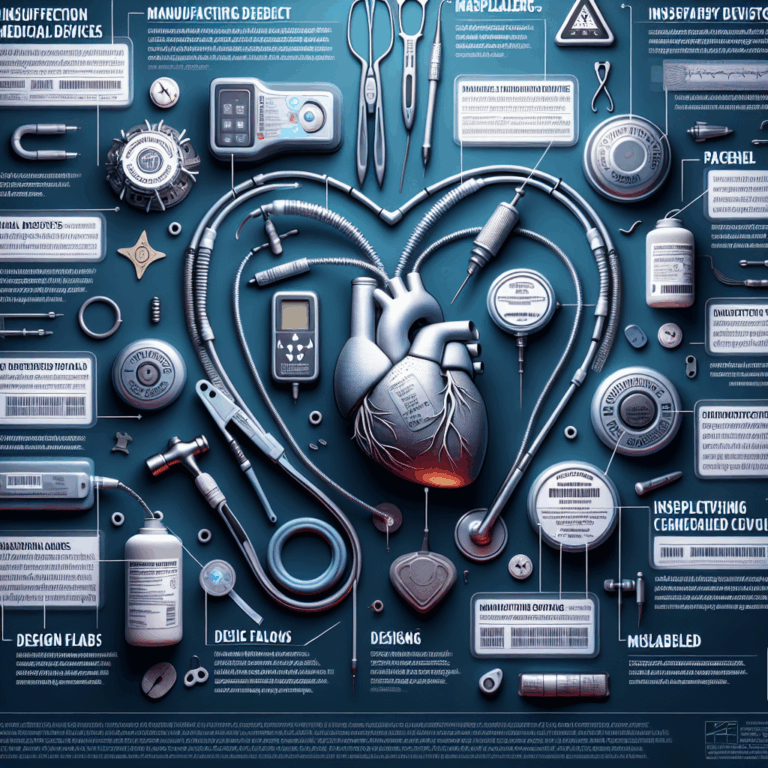
According to the U.S. Food and Drug Administration records, in an average year over 2,500 medical device recalls are issued in the United States. Some of these recalls simply require checking the device for problems, but others require the return or destruction of the device. Once identified, the FDA categorizes the root cause of these recalls into 40 categories, plus a catchall of “other”: situations that include labeling mix-ups, problems with expiration dates, and counterfeiting.
What’s shown here is the breakdown of the five biggest problem categories found among the 56,000 entries in the FDA medical-recall database, which stretches back to 2002: device design, process control (meaning an error in the device’s manufacturing process), nonconforming material/component (meaning something does not meet required specifications), software issues, and packaging.
Software issues are broken down into six root causes, with software design far and away the biggest problem. The other five are, in order: change control; software design changes; software manufacturing or deployment problems; software design issues in the manufacturing process; and software in the “use environment.” That last one includes cybersecurity issues, or problems with supporting software, such as a smartphone app.
This article appears in the December 2025 print issue as “Medical Device Recalls.”