Medical Device Recall
Procedure: Medical Device RecallCountry: Global Procedure Version #: 01/17/2024Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-MD-DRC-SRCategory: Medical DevicesValidity: Change, withdrawn, or NRA updateCertified…

Procedure: Medical Device RecallCountry: Global Procedure Version #: 01/17/2024Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-MD-DRC-SRCategory: Medical DevicesValidity: Change, withdrawn, or NRA updateCertified…

Procedure: Premarket Approval (PMA)Country: United StatesVersion #: 01/15/2024Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: US-MD-PMA-SRCategory: Medical DevicesValidity: Until MA expiration or next updateCertified…

Procedure: New Market Authorization Class I-IVCountry: Global ProcedureVersion #: 01/08/2024Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-MD-VI-IV-SRCategory: Medical DevicesValidity: For five years Certified…

Procedure: New Market Authorization Class IIICountry: Global ProcedureVersion #: 01/08/2024Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-MD-VIII-SRCategory: Medical DevicesValidity: For 5 years (US),…

Procedure: New Market Authorization Class IICountry: Global ProcedureVersion #: 01/08/2024Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-MD-VII-SRCategory: Medical DevicesValidity: For five years Certified…

Procedure: New Market Authorization Class ICountry: Global ProcedureVersion #: 01/08/2024Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-MD-VIA-SRCategory: Medical DevicesValidity: Until Change or local…

Procedure: The Breakthrough Devices ProgramCountry: United StatesVersion #: 01/18/2024Author: Vlad ReznikovEditor: Daria Kostiuchenko ID: US-MD-BDP-SRCategory: Medical DevicesValidity: Until 510(k), PMA or De…

Procedure: Pre-market Notification 510(k)Country: United StatesVersion #: 01/12/2024Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: US-MD-PMN-SRCategory: Medical DevicesValidity: Change, withdrawn, or FDA updateCertified by:…

Procedure: Q-Sub Activities Country: Global Procedure Version #: 01/03/2024 Author: Daria Kostiuchenko Editor: Daria Kostiuchenko ID: GP-MD-QSA-SR Category: Medical Devices Validity: Until MA Application Certified by: Vlad Reznikov Copyright: Pattern of USA Inc. SUMMARY Q-Submission (Q-Sub) refers to the important opportunities for submitters to share information with NRA and receive input outside of the submission…

Procedure: Q-Sub Activities Country: Global ProcedureVersion #: 01/03/2024Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-MD-QSA-SRCategory: Medical DevicesValidity: Until MA ApplicationCertified by: Vlad ReznikovCopyright:…

Procedure: Sunset Clause and MA closing activity Country: Global ProcedureVersion #: 01/05/2024Author: Vlad ReznikovEditor: Daria Kostiuchenko ID: GP-PhR-CSR-SRCategory: Pharmaceuticals/MD&IVDValidity: ImmediateCertified by: Vlad Reznikov Copyright: Pattern of USA Inc. SUMMARY A sunset clause refers to a regulatory provision that sets a predetermined expiration date or termination point for the marketing authorization of a product. This clause…

Procedure: Randomized Labeling for CRO Country: Global ProcedureVersion #: 01/05/2024Author: Vlad ReznikovEditor: Daria Kostiuchenko ID: GP-PhR-RND-SRCategory: Pharmaceuticals/MD&IVDValidity: Upon requestCertified by: Vlad ReznikovCopyright: Pattern of USA Inc. SUMMARY In drug development conducted by Contract Research Organizations (CROs), randomized labeling involves the assignment of unique codes or labels to investigational drug products in a random manner. This…

Procedure: Commercial Ad, Labeling and Health Claims Compliance Check Country: Global Procedure Version #: 01/05/2024 Author: Vlad Reznikov Editor: Daria Kostiuchenko ID: GP-PhR-CLH-SR Category: Pharmaceuticals/MD&IVD/Others Validity: Until next NRA update Certified by: Vlad Reznikov Copyright: Pattern of USA Inc. SUMMARY A Commercial Ad, Labeling, and Health Claims Compliance Check for healthcare products involves a thorough…

Procedure: Single Lay User Prototype UI/UX TestingCountry: Global ProcedureVersion #: 01/05/2024Author: Vlad ReznikovEditor: Daria Kostiuchenko ID: GP-MD-PTS-SRCategory: MD&IVD/OthersValidity: NACertified by: Vlad ReznikovCopyright: Pattern of USA Inc. SUMMARY Single Lay User Prototype UI/UX Testing is integral to the success of a healthcare startup developing medical devices. It aligns the design with user needs, identifies potential challenges,…

Procedure: NRA ClassificationCountry: Global ProcedureVersion #: 11/28/2023Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-PhR-CLS-SRCategory: PharmaceuticalsValidity: Until MA obtaining or NRA updateCertified by: Vlad ReznikovCopyright: Pattern of USA Inc. SUMMARY Divergent classification by national regulatory agencies is pivotal for companies to align with the distinct requirements of each target market. In the realm of regulatory affairs, it plays a crucial role…

Procedure: Authorized RepresentativeCountry: Global ProcedureVersion #: 11/30/2023Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-PhR-AR-SRCategory: PharmaceuticalsValidity: 1 year with optional extensionCertified by: Vlad ReznikovCopyright: Pattern of USA Inc. SUMMARY Legally Authorized Representative (AR, LAR, or US Agent in the United States) is a local person or legal entity appointed by foreign manufacturer to act on their behalf regarding certain regulatory obligations under local…
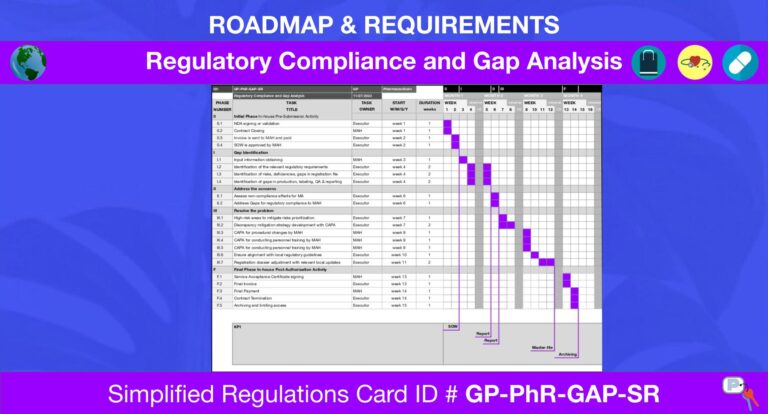
Procedure: Regulatory Compliance and Gap AnalysisCountry: Global ProcedureVersion #: 11/27/2023Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-PhR-GAP-SRCategory: PharmaceuticalsValidity: Until MA ApplicationCertified by: Vlad ReznikovCopyright: Pattern of USA Inc. SUMMARY Gap analysis involves assessing and identifying any shortcomings or gaps in a registration dossier compliance with local regulatory standards. These gaps could include missing documentation, incomplete testing, or lack of adherence to specific guidelines. Unlike risk…

Procedure: Intellectual Property Rights ProtectionCountry: Global ProcedureVersion #: 11/28/2023Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-PhR-IP-SRCategory: Pharmaceuticals/MD&IVD/OthersValidity: IP Rights Expiration DateCertified by: Vlad ReznikovCopyright: Pattern of USA Inc. SUMMARY Intellectual Property Rights Protection in pharmaceuticals registrations refers to the legal mechanisms and regulations in place to safeguard the intellectual property of pharmaceutical companies. This includes patents, trademarks, copyrights, and trade secrets…

Procedure: Regulatory Intelligence Country: Global Procedure Version #: 11/28/2023 Author: Daria Kostiuchenko Editor: Daria Kostiuchenko ID: GP-PhR-RI-SRC Category: Pharmaceuticals/MD&IVD/Others Validity: Until next NRA update Certified by: Vlad Reznikov Copyright: Pattern of USA Inc. SUMMARY Regulatory Intelligence involves up-to-date gathering, analysis, and interpretation of information regarding regulations and compliance requirements. Strategic consulting involves providing expert advice…