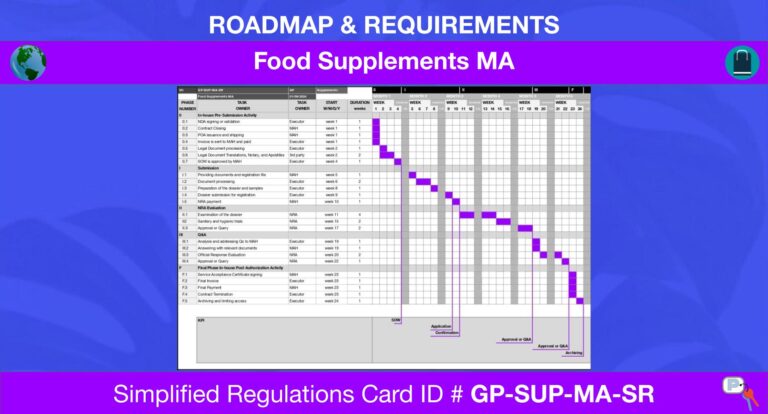
MA Renewal
Procedure: MA RenewalCountry: Global ProcedureVersion #: 01/17/2024Author: Daria KostiuchenkoEditor: Daria Kostiuchenko ID: GP-MD-PhR-SRCategory: Pharmaceuticals/MD&IVDValidity: Lifetime in most countriesCertified by: Vlad Reznikov Copyright: Pattern of USA Inc. …