Abhishek Appaji has committed his career to bringing lifesaving technology to underresourced communities. The IEEE senior member weaves together artificial intelligence, biomedical engineering, deep learning, and neuroscience to make doctors’ jobs easier and to improve patient outcomes.
“The intersection of these fields is where the most impactful breakthroughs in diagnostic precision occur,” says Appaji, an associate professor of medical electronics engineering at the B.M.S. College of Engineering, in Bengaluru, India.
Abhishek Appaji
Employer
B.M.S. College of Engineering, in Bengaluru, India
Job title
Associate professor of medical electronics engineering
Member grade
IEEE senior member
Alma maters
B.M.S. College of Engineering; University of Visvesvaraya, in Bengaluru; Maastricht University, in the Netherlands
Many of his inventions have been deployed in remote areas of India, providing physicians with quality diagnostic tools, including an AI-powered machine that can scan retinas to detect medical conditions and a smart bed that continuously monitors a patient’s vital signs.
An active volunteer with the IEEE Young Professionals Bangalore Section, he has launched professional networking events, technology workshops, a mentorship program, and other initiatives.
For his “contributions to accessible AI-driven health care solutions and leadership in empowering young professionals,” Appaji is the recipient of this year’s IEEE Theodore W. Hissey Outstanding Young Professional Award. The honor is sponsored by the IEEE Photonics and Power & Energy societies as well as IEEE Young Professionals. The award is scheduled to be presented this month during the IEEE Honors Ceremony in New York City.
“This award represents a significant milestone in my career,” Appaji says. “It validates my core belief that our success as engineers is not solely measured by research outcomes or publications but by the tangible impact we have on lives through accessible technology and the quality of the next generation of leaders we empower.”
Developing a blood glucose measurement device
After earning a bachelor’s degree in engineering from B.M.S. in 2010, he joined the school as a lecturer in its medical electronics engineering department. At the same time, he pursued master’s degrees in bioinformatics at the University Visvesvarya College of Engineering, also in Bengaluru. He graduated in 2013 and continued to teach at B.M.S.C.E.
Four years later, Appaji signed up for the MIT Global Entrepreneurship Bootcamp, a two-week intensive hybrid program that includes webinars, online courses, and a five-day stay at MIT. It’s designed to give teams of aspiring entrepreneurs, innovators, and early-stage founders the structured mindset, tools, and frameworks they need to succeed.
Appaji says he discovered the program while researching opportunities in innovation.
“I had the technical expertise, but I needed a structured framework to transition my research from the laboratory to the market,” he says.
During the MIT boot camp, he and a team of four other participants were tasked with approaching a complex health care challenge. They developed a noninvasive blood glucose measurement device to manage gestational diabetes—a condition that causes high blood sugar and insulin resistance during pregnancy. When the program ended, Appaji and two of his Australia-based teammates continued their collaboration by founding Glucotek in Brisbane, Australia.
Inspired to continue his research in health care technology, Appaji pursued a doctorate in mental health and neurosciences at Maastricht University, in the Netherlands.
His thesis focused on computational methods to identify retinal vascular patterns.
“The patterns we analyze—including the curvature of the vessels, the angles at which they branch out, and their dimensions—reveal the health of the microvascular system,” he says. “With conditions like schizophrenia and bipolar disorder, microvascular changes mirror neurovascular changes in the brain.”
“My journey has shown me that IEEE is much more than a professional society; it is a global platform that allows me to collaborate with a diverse network of experts to solve local humanitarian challenges.”
Examining and measuring the retinal vascular system offers physicians a noninvasive way to examine neural changes, which can be biomarkers for psychiatric illnesses, he says.
To bring his idea to life, he collaborated with an ophthalmologist, a psychiatrist, and colleagues from his engineering school to develop a screening device. They also created and trained the AI models that analyze retinal images.
Ideas from his thesis led to the creation of the Smart Eye Kiosk, an AI-powered tool that scans the network of small veins that deliver blood to the inner retina. The tool monitors stress levels and mental health. It also screens for basic eye diseases such as diabetic retinopathy, as well as damage to retinal blood vessels caused by high blood sugar.
Retinal images also can reveal physiological changes in the brain associated with psychiatric disorders such as schizophrenia and bipolar disorder, Appaji says. The kiosk uses AI models to analyze measurements of the vasculature network, such as vessel thickness, which can be biomarkers for psychiatric conditions. Since mental illnesses can be linked to genetics, relatives of patients with schizophrenia and bipolar disorder were also invited to participate in a study funded by India’s Cognitive Science Research Initiative’s Department of Science & Technology. The clinical data from this study can pave the way for earlier, more accurate diagnoses.
“The biological basis for this is fascinating,” Appaji says. “The retina is the only place in the human body where the central nervous system and the vascular system can be visualized directly and noninvasively. Anatomically, the retina is an extension of the posterior part of the brain. Therefore, physiological changes in the brain are often reflected in the eyes.”
This kiosk was developed in collaboration with Tan Tock Seng Hospital and Nanyang Technological University, which was funded by Ng Teng Fong Healthcare Innovation Program.
He earned his Ph.D. in 2020 from Maastricht, and he received the Best Thesis Award from the university’s Mental Health and Neuroscience Research Institute. Appaji credits his time at the school for his multidisciplinary approach to developing medical devices.
“Having the perspectives of mentors from diverse fields was essential to help me move my research beyond theory into a data-driven diagnostic tool,” he says.
He was then named institutional coordinator of R&D at B.M.S. and later was promoted to be its head.
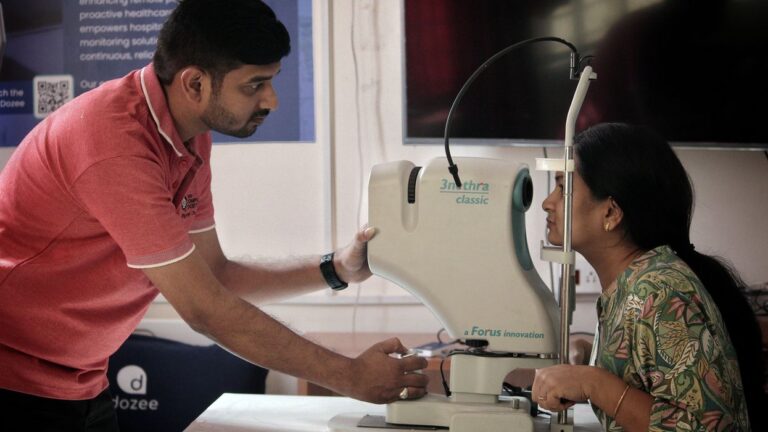
 Abhishek Appaji working on a smart bed sensor that continuously monitors a patient’s vital signs without the use of wires or wearable sensors.Abhishek Appaji
Abhishek Appaji working on a smart bed sensor that continuously monitors a patient’s vital signs without the use of wires or wearable sensors.Abhishek Appaji
A wireless smart bed to monitor vital signs
Appaji continues to develop technologies for patients who need them most. “I feel a deep need to bridge this gap and ensure innovations have a tangible impact on society,” he says. In addition to the Smart Eye Kiosk, he improved the performance of the sensors of the smart beds that continuously monitor a patient’s vital signs without the use of wires or wearable sensors. The beds help hospital staff check on their patients in a noninvasive way.
The project was done in collaboration with health AI company Dozee (Turtle Shell Technologies) in Bengaluru. The system measures mechanical microvibrations produced by the body in response to the ejection of blood into the aorta, which occurs with each heartbeat. A thin, industrial-grade sensor sheet is placed underneath the mattress. Additional funding is being provided by India’s Department of Science and Technology.
“These sensors are incredibly sensitive,” Appaji says. “They pick up minute mechanical tremors through the mattress material.”
The sensors detect the force of the patient’s heartbeat and the expansion and contraction of their chest during respiration. The vibrations are converted into electrical signals and analyzed using deep learning algorithms developed by Appaji and his team at the university in collaboration with Dozee.
The technology is used in more than 200 hospitals throughout India and in thousands of households, he says.
Mentoring budding entrepreneurs
Appaji is also executive director of the BMSreenivasiah Innovators Guild Foundation, dedicated to nurturing entrepreneurial talent among students and faculty across the BMS group of Institutions. A not-for-profit company promoted by the BMS Education Trust, BIG Foundation provides a structured ecosystem for innovation, incubation, and startup growth.
There, Appaji mentors budding entrepreneurs, offering advice on business plans, product pitches, marketing strategies, and licensing. Participants are students and faculty members.
The foundation has incubated more than 10 ventures, according to Appaji.
“The majority are centered on health care applications,” he says, “and have successfully secured backing from investors and seed funds.”
Taking IEEE’s mission to heart
Appaji was introduced to IEEE as an undergraduate when one of his professors encouraged him to volunteer for a conference sponsored by the IEEE Engineering in Medicine and Biology Society. He transcribed the seminars for session chairs, assisted with managing the talks, and helped answer attendees’ questions.
“That experience was transformative,” he recalls. “I was amazed to find myself in the same room with the speakers and scientists who had authored the very textbooks I was studying.
“It was then that I realized IEEE is far more than just technology and volunteering; it is a global platform for high-level networking with world-class scientists and technologists.”
Appaji has served in several IEEE leadership positions, including 2018–2019 chair of the Young Professionals Bangalore Section. He is now treasurer of the IEEE Education Society, chair of IEEE Computer Society Bangalore Chapter, member of the steering committee of IEEE DataPort, and serves on the IEEE Member and Geographic Activities and IEEE Educational Activities boards.
“What motivates me to remain active within IEEE is the profound alignment between my personal goals and the organizational mission of advancing technology for the benefit of humanity,” he says. “My journey has shown me that IEEE is much more than a professional society; it is a global platform that allows me to collaborate with a diverse network of experts to solve local humanitarian challenges.”
The organization has helped fund some of Appaji’s lifesaving work. During the COVID-19 pandemic, he received a grant from the IEEE Humanitarian Technologies Board and Region 10 to develop 3D-printed protective equipment for people in Bengaluru’s underserved communities. The virus spread quickly in the high-density areas, where social distancing was nearly impossible. The kits, which included a door opener to avoid high-touch surfaces and an elbow-operated soap dispenser, were sent to nearly 500 households.
“This work remains one of my most meaningful contributions to humanitarian technology,” Appaji says, “demonstrating how engineering can be rapidly deployed to protect vulnerable populations during a global crisis.”
He advises younger IEEE members to: “Say yes to taking on roles of responsibility. Don’t wait for a formal title to lead; instead, start by volunteering to do small, manageable tasks within your local chapter or section.”
“The networking opportunities and leadership skills you gain through these early responsibilities will shape your professional career far more than any textbook ever could.”






























 Abhishek Appaji working on a smart bed sensor that continuously monitors a patient’s vital signs without the use of wires or wearable sensors.Abhishek Appaji
Abhishek Appaji working on a smart bed sensor that continuously monitors a patient’s vital signs without the use of wires or wearable sensors.Abhishek Appaji